Service Offerings
Service Offerings Quick Guide
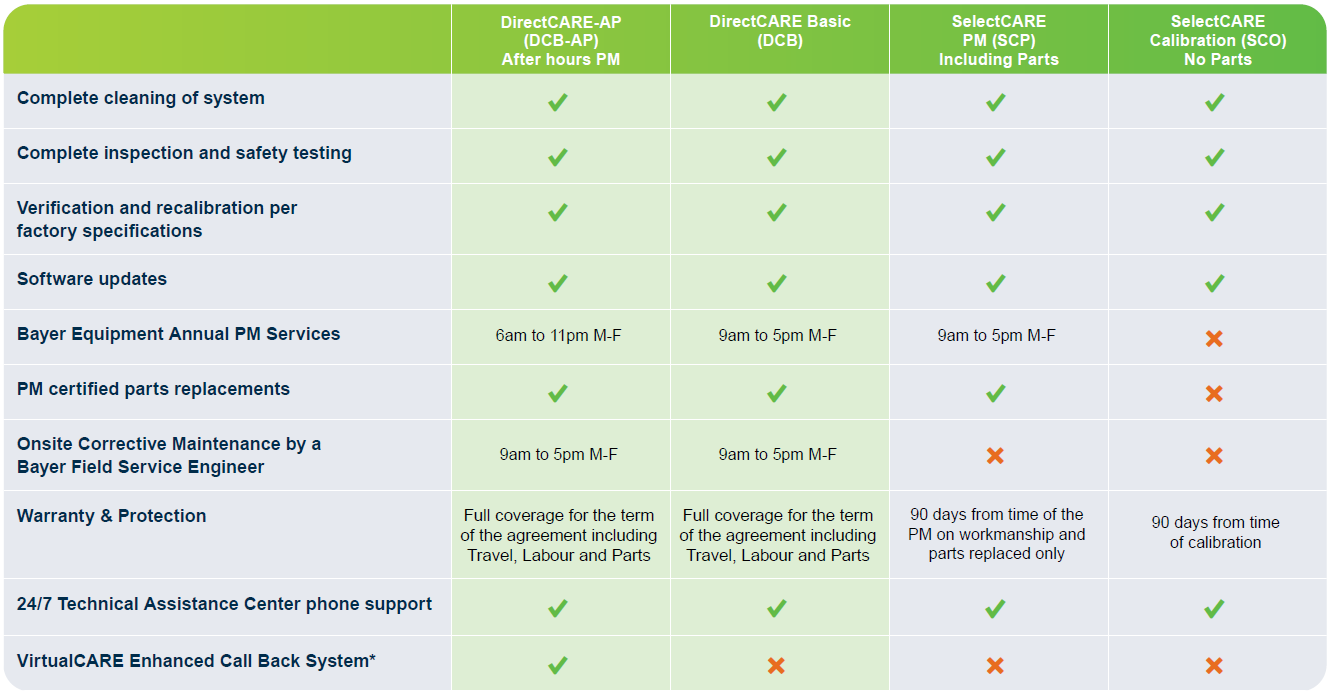
*Only applies to Bayer Equipment with VirtualCARE functionality
Learn More
Want to Know More?

Have VirtualCARE but need help requesting a service request?
Contact Us
Service Offerings

*Only applies to Bayer Equipment with VirtualCARE functionality
Learn More

Have VirtualCARE but need help requesting a service request?
Contact Us