What is Ultravist® ?
Ultravist® is an iodine-based non-ionic monomeric low osmolar extracellular X-ray contrast medium (LOCM). It can be used for X-ray techniques requiring contrast enhancement such as conventional radiography, angiography and computed tomography.1
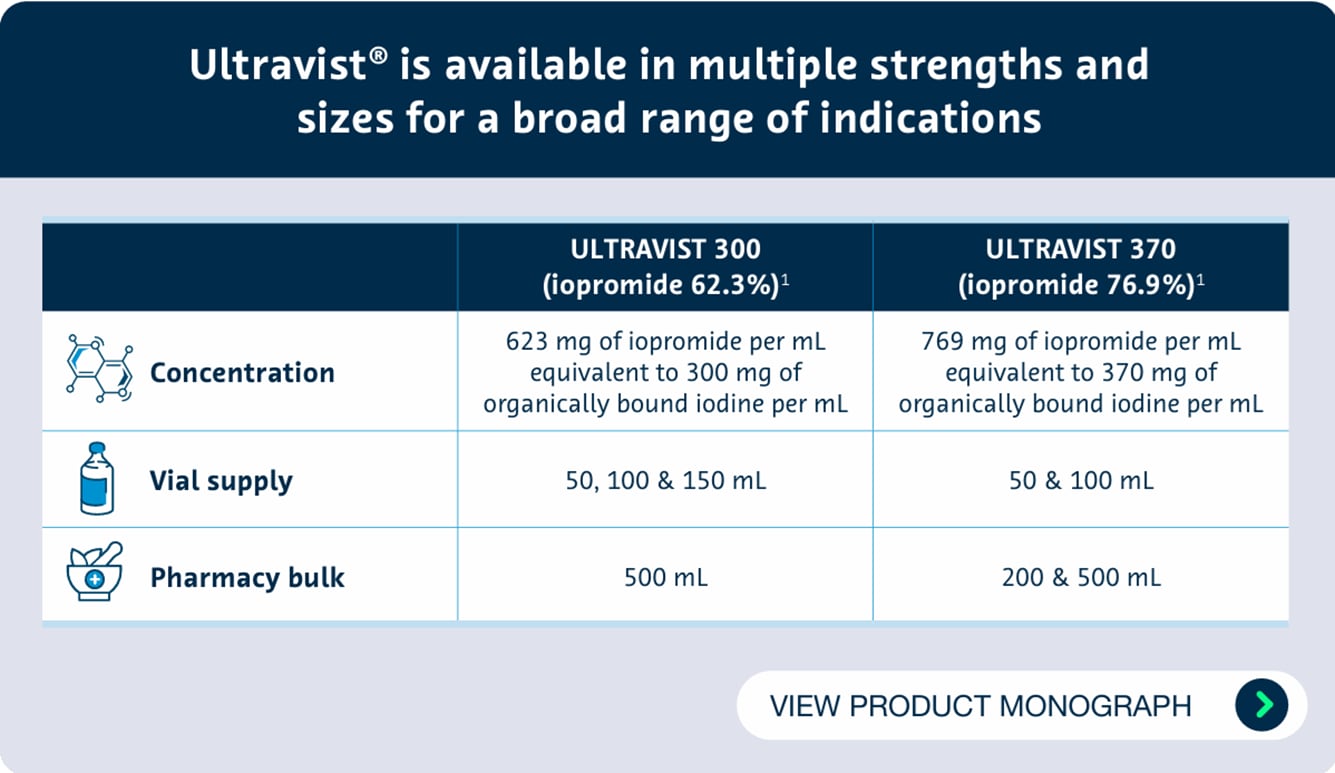
ULTRAVIST (iopromide), provided as 300 mg I/mL and 370 mg I/mL, is indicated for:
- Intravascular use to provide diagnostic information in a number of radiographic contrast procedures.
- Visualization of various body cavities, e.g., arthrography and hysterosalpingography.
- ULTRAVIST 300: computed tomography (CT), excretory urography, pediatric excretory urography, renal arteriography, peripheral arteriography (bifemoral pelvis/leg), cerebral arteriography, phlebography of the extremities, arthrography
- ULTRAVIST 370: computed tomography (CT), excretory urography, coronary arteriography (including PTCA) with or without left ventriculography. pediatric angiocardiography, arthrography
- Differences in safety may be seen in geriatric patients (≥65 years of age)
Consult the Product Monograph at https://www.bayer.com/sites/default/files/2020-11/ultravist-pm-en.pdf for important information on:
- Contraindications for use in myelography, cerebral ventriculography, and cisternography; patients with manifest hyperthyroidism; intrathecal use; and preparatory dehydration in pediatric patients.
- Other relevant warnings and precautions regarding: blood clotting; serious and fatal reactions; obscuring of lesions seen on prior unenhanced CT scans; increased radiation exposure; patients with cardiac and circulatory insufficiency, hypertension, pheochromocytoma, cerebral arteriosclerosis, latent hyperthyroidism, severe impairment of hepatic or renal function, pulmonary emphysema, diabetes with renal dysfunction requiring treatment, cerebral arterial spasm, bland nodular goiter, multiple myeloma or other paraproteinemias, and renal transplant; risk of thrombosis and embolism with angiography in homocystinuria; patients with pronounced states of excitement, anxiety, and pain; patients with congestive heart failure receiving concurrent diuretic therapy; changes in thyroid function; low thyroid hormones in infants exposed during pregnancy or after; obscuring of thyroid test results; patients with sickle cell disease; patients undergoing oral cholecystography; patients with a history of allergy, sensitivity to iodine/radiographic media, previous reaction to contrast media, and bronchial asthma; hemodynamic disturbances; inadvertent injection into an aortic branch or arterial trunk; risks of organ damage, nerve injuries, thromboembolic events, and vascular injury during angiographic procedures; specialized personnel and monitoring requirements for coronary arteriography and ventriculography; patients suspected of having thromboangilitis obliterans; patients with increased intracranial pressure, disrupted bloodbrain barrier or condition that prolongs contrast material in vessels; hemorrhage from site of arterial puncture; patients with a reduced seizure threshold or neurologic disorders; neurological complications are more frequent in cerebral angiography and related procedures; patients with myasthenia gravis; patients with renal insufficiency; pregnancy and breastfeeding; and pediatric patients with asthma, sensitivity to medication/allergens, heart disease, congestive heart failure or serum creatinine > 1.5 mg/dL.
- Conditions of clinical use, adverse reactions, drug interactions, and dosing instructions.
The Product Monograph is also available by calling Bayer Medical Information at 1-800-265-7382.
Delivering Confidence with Every Dose
Backed by more than 150,000 patients in observational studies, Ultravist® is well established in clinical research.3-5
Fewer delayed skin reactions
In a German study, delayed skin reactions occurred significantly less often with monomeric Ultravist® than with the dimeric agent iotrolan (9.7% vs 16.4%, p<0.05). An Iranian study showed a similar pattern: late reactions such as itching and rash were more frequent with the iso-osmolar iodixanol (7.9% and 14.3%) than with LOCM Ultravist® (3.5% and 1.8%, p<0.01).7,8†‡
German Study
Iranian Study
HSR = hypersensitivity reaction
* This was a multicenter, retrospective, non-randomized observational study conducted at tertiary hospitals in South Korea from January 2013 to December 2017, including 196,081 patients who received iodinated contrast media during radiologic procedures to evaluate hypersensitivity reactions.
† This was a single center, prospective, single-blinded, randomized controlled trial conducted in Germany between 1996 and 2002. The study included 895 patients who either received iotrolan (n=300), Ultravist® (n=297), or underwent unenhanced CT (n=298) to compare adverse events across these patient populations.
‡ This was a single center, prospective, randomized study of 120 patients at an Iranian hospital who received either LOCM Ultravist® or iso-osmolar iodixanol to undergo abdominopelvic CT scans, with the aim to compare the allergic adverse effects of each contrast agent
§ Data were analyzed from Bayer’s pharmacovigilance database, covering >288 million Ultravist® injections worldwide (January 1995–December 2020), with exposure estimated from sales and market-research data. The analysis included all ages and all indications and primarily assessed the risk of HSRs in children (<18 years) and elderly patients (≥65 years).
¶ This was an analysis of 152,233 patients from 4 observational studies with the objective of analyzing the potential impact of patients’ sex, race, and region/country on the risk of HSRs after intravenous or intraarterial administration of Ultravist®.
Dedication to Quality
Let’s build the future with confidence
Every step of the Ultravist® production process follows rigorous quality standards that include GMP
(Good Manufacturing Practices) and ISO 9001, ensuring the highest quality output.
The manufacturing process – which begins in Berlin, Germany – includes
Continuous quality control of materials and packaging
Process control at every step
Dedication to improving the ecological footprint across our operations and the entire value chain